Have you ever wondered what happens when proteins inside your cells don’t fold properly?
In this session, let’s explore the Unfolded Protein Response (UPR) and Endoplasmic Reticulum (ER) Stress — two vital processes that decide whether a cell survives or dies when misfolded proteins accumulate.
Normal Protein Folding: The Role of Chaperones
Inside the endoplasmic reticulum (ER), newly formed proteins enter the lumen, where chaperone proteins help them fold into their correct 3-D shapes.
Properly folded proteins then leave the ER to perform their normal functions.
This is how healthy protein synthesis and folding maintain cellular balance.
What Causes Protein Misfolding?
Several factors can disturb this delicate process:
-
Metabolic alterations
-
Mutations in proteins or chaperones
-
Viral infections causing excess viral protein synthesis
-
Chemical injury
-
ATP depletion (lack of energy for proper folding)
-
Changes in pH or redox state
-
Aging
-
Diseases like plasma cell tumors or insulin resistance
When any of these occur, misfolded proteins start piling up, creating stress inside the ER.
What Is ER Stress?
ER stress happens when the protein folding demand exceeds the folding capacity of the endoplasmic reticulum.
In simple terms, the cell is trying to fold more proteins than it can handle.
This imbalance leads to accumulation of misfolded proteins inside the ER lumen — a key trigger for cellular stress.
How Does the Cell Detect ER Stress?
The ER has sensor proteins on its surface that can sense misfolded proteins.
When activated, these sensors send signals into the cytoplasm to start a defense mechanism called the Unfolded Protein Response (UPR).
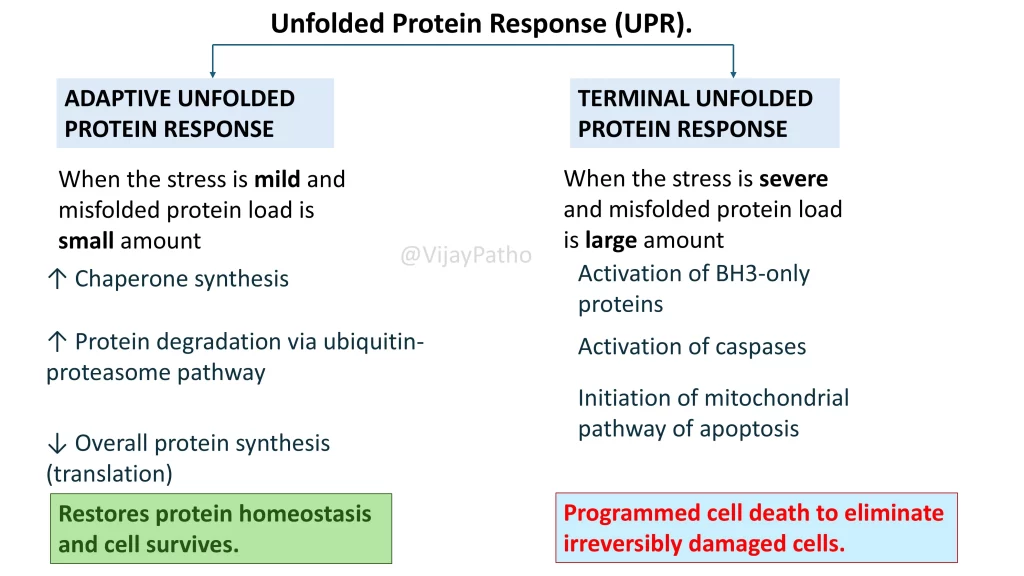
Unfolded Protein Response (UPR): The Two Pathways
Once the UPR is activated, the cell chooses between two outcomes — adaptation or death, depending on how severe the stress is.
1. Adaptive UPR (Cell Survival)
When the stress is mild, the cell tries to recover by:
-
Producing more chaperones to assist folding
-
Degrading abnormal proteins through the ubiquitin-proteasome pathway
-
Reducing overall protein synthesis to lower the load
These steps restore protein balance (protein homeostasis) and help the cell survive.
2. Terminal UPR (Cell Death)
When stress becomes severe and the buildup of misfolded proteins is overwhelming:
-
BH3-only proteins and caspases are activated
-
The mitochondrial apoptosis pathway is triggered
-
The cell undergoes programmed cell death (apoptosis)
This is the body’s way of removing damaged cells that cannot recover.
Diseases Linked to Protein Misfolding
| Disease | Affected Protein | Mechanism / Effect |
|---|---|---|
| Cystic Fibrosis | CFTR | Loss of chloride transport |
| Familial Hypercholesterolemia | LDL receptor | Defective cholesterol uptake |
| Tay-Sachs Disease | Hexosaminidase-β | GM2 ganglioside accumulation in neurons |
| α₁-Antitrypsin Deficiency | α₁-Antitrypsin | Liver apoptosis; emphysema in lungs |
| Alzheimer’s Disease | Aβ peptide | Aggregation leading to neuronal apoptosis |
Each of these disorders shares a common mechanism — misfolded proteins and ER stress.
Clinical Significance of ER Stress and UPR
Understanding ER stress is crucial because it plays a role in many conditions:
-
Neurodegenerative diseases such as Alzheimer’s and prion diseases
-
Viral infections that overload the ER with viral proteins
-
Diabetes, due to excessive insulin synthesis
-
Ischemia and hypoxia, where ATP depletion hampers folding
-
Cancers, where tumor cells constantly produce excess proteins
Whether a cell adapts or dies depends on how effectively it manages ER stress.
Summary
The Unfolded Protein Response (UPR) acts as a cellular quality-control system.
When proteins misfold, the ER activates this mechanism to restore balance.
If the stress is manageable, the cell survives. If not, apoptosis eliminates the damaged cell.
This process explains how protein misfolding contributes to diseases like cystic fibrosis, diabetes, and Alzheimer’s disease — and highlights how cells constantly walk a fine line between survival and death.
CLICK below to watch the video tutorial on ER Stress & Unfolded Protein Response
Click to learn about Integrated stress response