Polycythemia Vera Simplified: JAK2 Mutation, Pathogenesis, Morphology & Diagnosis
What Is Polycythemia Vera?
Polycythemia Vera is a chronic myeloproliferative disorder in which the bone marrow produces too many red blood cells, along with an increase in white cells and platelets — a condition known as panmyelosis.
Among these, the rise in red blood cell mass is the most prominent and is responsible for most of the clinical features.
Relative vs. Absolute Polycythemia
Understanding this distinction is important:
-
Relative Polycythemia – Caused by reduced plasma volume, usually due to dehydration. The red cell mass remains normal.
-
Absolute Polycythemia – Due to an actual increase in red cell mass.
-
Primary Absolute Polycythemia – Seen in Polycythemia Vera (bone marrow abnormality).
-
Secondary Absolute Polycythemia – Due to increased erythropoietin (EPO) production, as in hypoxia or certain tumors (paraneoplastic syndromes).
-
Pathogenesis Polycythemia Vera
The key molecular abnormality in PV is a mutation in the JAK2 gene on chromosome 9p.
JAK stands for Janus Kinase, a non-receptor tyrosine kinase protein involved in signal transduction.
To understand this, let’s recall the JAK-STAT pathway:
-
Growth factors like erythropoietin (EPO), thrombopoietin, or G-CSF bind to their receptors on hematopoietic stem cells.
-
This activates the JAK2 protein, which then phosphorylates and activates STAT proteins.
-
The activated STAT dimers move to the nucleus and stimulate genes responsible for cell survival and proliferation.
In PV, a point mutation (V617F) replaces valine with phenylalanine at position 617 of the JAK2 protein.
This makes the pathway constantly active, even without erythropoietin. The cells keep dividing independently — leading to excessive blood cell production.
Types of JAK2 Mutations
-
Heterozygous Mutation – Only one copy of the JAK2 gene is mutated. Seen in most patients (~70%).
-
Homozygous Mutation – Both copies mutated (25–30% cases), due to duplication or mitotic recombination.
The homozygous state causes more severe disease, faster progression to marrow fibrosis (the “spent phase”), and features like splenomegaly and itching.
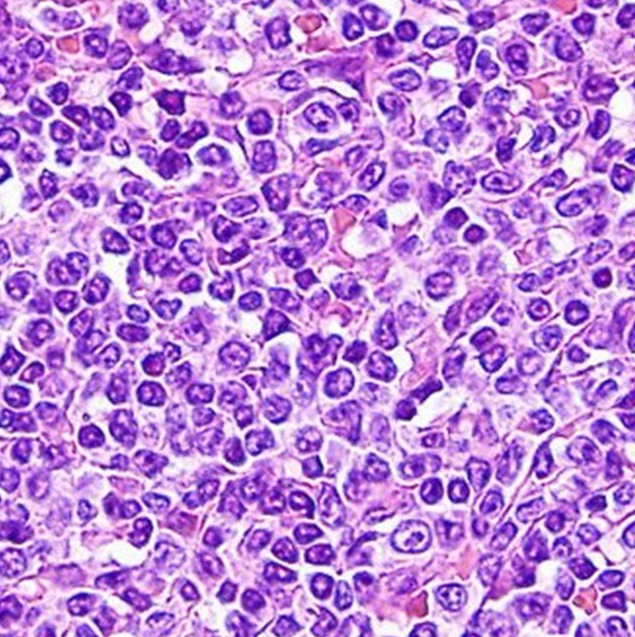
Morphology of Polycythemia Vera
Peripheral Blood
-
Hematocrit: Markedly increased → thick, viscous blood.
-
Smear: Crowded red cells with some distortion.
-
Cells: Usually normocytic normochromic, but may become microcytic hypochromic if iron deficiency develops.
-
Leukocytosis: Neutrophilia with left shift.
-
Basophilia: Common finding.
-
Thrombocytosis: Large, often abnormal platelets.
Bone Marrow
-
Hypercellular marrow with trilineage growth (erythroid, myeloid, and megakaryocytic proliferation).
-
Reticulin fibrosis may be seen early in about 10% of cases.
-
In advanced stages (“spent phase”), marrow fibrosis becomes extensive and hematopoietic cells shift to extramedullary sites like spleen and liver.
Clinical Features of Polycythemia Vera
Most symptoms stem from increased blood viscosity and reduced blood flow:
-
Plethoric and cyanotic appearance due to sluggish circulation
-
Headache, dizziness, and hypertension
-
Pruritus (itching), especially after a warm bath — due to histamine release from basophils
-
Gastrointestinal complaints and peptic ulceration
-
Hyperuricemia and gout (from increased cell turnover)
-
Thrombotic events: Deep vein thrombosis, myocardial infarction, stroke, hepatic vein thrombosis (Budd-Chiari syndrome), and portal vein thrombosis
-
Bleeding tendencies such as epistaxis or gum bleeding may also occur
Diagnostic Criteria (WHO) of Polycythemia Vera
Diagnosis requires either all three major criteria or the first two major plus one minor criterion:
Major Criteria
-
Elevated Hemoglobin / Hematocrit
-
16.5 g/dL in men, >16 g/dL in women
-
or hematocrit >49% in men, >48% in women
-
-
Bone Marrow Hypercellularity
-
Trilineage (panmyelosis) proliferation, adjusted for age
-
-
Presence of JAK2 Mutation
-
V617F or exon 12 mutation
-
Minor Criterion
-
Subnormal Serum Erythropoietin Level
Treatment and Prognosis
-
Phlebotomy is the mainstay — maintaining near-normal red cell mass to reduce symptoms and complications.
-
JAK2 inhibitors exist but are not yet highly effective compared to BCR-ABL inhibitors used in CML.
-
Prognosis:
-
About 15–20% develop myelofibrosis within 10 years.
-
Around 2% may progress to acute myeloid leukemia (AML), rarely to lymphoblastic leukemia.
-
Click below to watch the video on youtube
You can learn more on other myeloproliferative disorder Chronic Myeloid Leukemia here