Chronic Lymphocytic Leukemia (CLL) – Epidemiology, Pathogenesis, Morphology, Clinical Features
What is Chronic Lymphocytic Leukemia (CLL)?
-
CLL is a peripheral B-cell neoplasm and the most common leukemia in adults in the Western world.
-
It has a closely related counterpart called Small Lymphocytic Lymphoma (SLL).
-
The difference lies mainly in the blood counts:
-
CLL shows absolute lymphocytosis (>5000/mm³) in peripheral blood.
-
SLL is primarily a lymph node–based neoplasm (a type of Non-Hodgkin lymphoma) without significant blood lymphocytosis.
-
Epidemiology
-
Age: Common around 60 years.
-
Gender: More common in males than females.
-
SLL accounts for only ~4% of all non-Hodgkin lymphomas.
Pathogenesis – How Does Chronic Lymphocytic Leukemia (CLL) Develop?
Unlike many other lymphoid malignancies, chromosomal translocations are rare in CLL. Instead, certain chromosomal deletions and mutations play key roles.
1. Chromosomal Abnormalities
-
Most common: Deletion of long arm of chromosome 13 (del13q)
-
Others: del11q, del17p, and trisomy 12
2. Role of microRNAs
-
The deleted 13q region encodes microRNA-15A and microRNA-16-1, which act as tumor suppressors.
-
Their loss leads to overexpression of BCL2 (an anti-apoptotic protein) → reduced apoptosis → accumulation of long-lived lymphocytes.
3. Somatic Hypermutation Status
-
Normally occurs in germinal center B cells to improve antibody affinity.
-
Mutated immunoglobulin genes (with somatic hypermutation): indicate origin from post–germinal center memory B cells → better prognosis
-
Unmutated genes (no somatic hypermutation): indicate naive B-cell origin → more aggressive disease
4. NOTCH1 and RNA Splicing Mutations
-
NOTCH1 gain-of-function mutations seen in 10–18% cases → associated with poor prognosis
-
RNA splicing gene mutations also occur.
Summary of genetic changes:
-
del13q → ↓miR15/16 → ↑BCL2 → ↓apoptosis → lymphocyte accumulation
-
Unmutated Ig genes → aggressive; Mutated Ig genes → better outcome
-
NOTCH1 and RNA splicing mutations → poor prognosis
Tumor Microenvironment and Growth
-
Proliferation centers in lymph nodes are key growth zones.
-
Stromal cells in these centers release:
-
NF-κB → promotes cell survival
-
MYC → promotes proliferation
-
-
B-cell receptor (BCR) signaling pathway via BTK (Bruton Tyrosine Kinase) is crucial.
-
BTK inhibitors like Ibrutinib block this pathway and show good therapeutic response.
-
Morphology of Chronic Lymphocytic Leukemia (CLL)
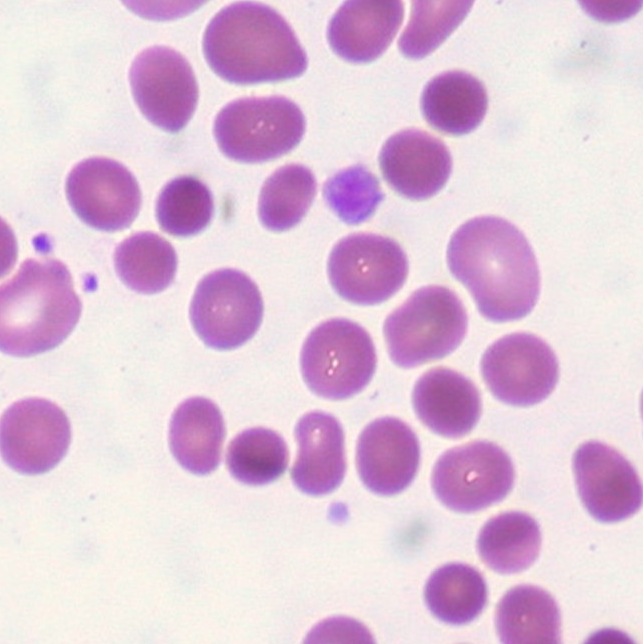
Peripheral Blood
-
Numerous small lymphocytes with:
-
Dense chromatin
-
Very scant cytoplasm
-
-
Smudge cells (basket cells) — fragile nuclei crushed during smear preparation.
-
Highly characteristic, but not specific (can appear in other conditions too).
-
Lymph Node (SLL)
-
Diffuse effacement of nodal architecture (no cortex/medulla)
-
Sheets of small lymphocytes
-
Proliferation centers — loose aggregates of larger activated lymphocytes
Immunophenotype
CLL/SLL cells express:
-
Pan B-cell markers: CD19, CD20
-
CD23 and CD5 co-expression
-
Low-level surface immunoglobulin (usually IgM, sometimes IgD/IgG/IgT)
-
High BCL2 expression
Clinical Features of Chronic Lymphocytic Leukemia (CLL)
-
Often asymptomatic at diagnosis
-
Symptoms, if present:
-
Fatigue, weight loss, anorexia
-
Generalized lymphadenopathy
-
Hepatosplenomegaly (in 50–60% cases)
-
Immune Dysfunction
-
Despite high lymphocyte counts, normal immune function is disrupted
-
Hypogammaglobulinemia → increased bacterial infections
-
Autoantibodies → autoimmune hemolytic anemia or thrombocytopenia (10–15% cases)
Staging of Chronic Lymphocytic Leukemia (CLL)
There are two staging systems:
Rai Classification (Used in USA)
-
Stage 0: Lymphocytosis (>10,000/mm³ or >30% marrow lymphocytes)
-
Stage I: Stage 0 + lymphadenopathy
-
Stage II: Stage 0 + hepatomegaly or splenomegaly
-
Stage III: Stage 0 + anemia (Hb <11 g/dL)
-
Stage IV: Stage 0 + thrombocytopenia (platelets <1 lakh/mm³)
-
Stages III and IV represent advanced disease (marrow infiltration suppressing hematopoiesis).
Binet Classification (Used in Europe)
-
Stage A: Lymphocytosis; Hb >10 g/dL; Platelets >1 lakh/mm³; <2 involved sites
-
Stage B: Same as A, but 3–5 involved sites
-
Stage C: A/B + Hb <10 g/dL or platelets <1 lakh/mm³
Involved sites include cervical, axillary, inguinal nodes, liver, and spleen.
Treatment of Chronic Lymphocytic Leukemia (CLL)
-
Evolving; multiple modalities:
-
BCL2 inhibitors
-
Anti-CD20 antibodies
-
BTK inhibitors (e.g. Ibrutinib)
-
-
These are chosen based on stage, age, and genetic profile.
Prognosis of Chronic Lymphocytic Leukemia (CLL)
-
Median survival ~10 years
-
Worse prognosis with:
-
Advanced stage (Rai III/IV or Binet C)
-
del11q, del17p (TP53)
-
Unmutated Ig genes
-
NOTCH1 mutation
-
ZAP70 expression (enhances Ig receptor signaling)
-
Richter Transformation in Chronic Lymphocytic Leukemia (CLL)
-
In 5–10% cases, CLL/SLL can transform to Diffuse Large B-Cell Lymphoma (DLBCL)
-
Known as Richter Syndrome
-
Presents as rapidly enlarging lymph node or splenic mass
-
Involves additional TP53 and MYC mutations
-
Has a very poor prognosis
Key Takeaways
-
CLL is the most common adult leukemia
-
Originates from B-cells with various genetic alterations and microenvironmental support
-
Shows smudge cells in blood and proliferation centers in lymph nodes
-
Immune dysfunction is common despite high lymphocyte counts
-
Stage and genetic profile determine prognosis
-
Richter transformation is a dangerous complication
CLICK HERE to view the video tutorial on chronic lymphocytic leukemia