Acute Lymphoblastic Leukemia (ALL) – Simplified
What is Acute Lymphoblastic Leukemia (ALL)?
ALL (also called acute lymphoblastic lymphoma in some cases) is a neoplasm of immature B or T cells.
-
These immature cells are called lymphoblasts.
-
They may be B-lymphoblasts or T-lymphoblasts.
Who gets Acute Lymphoblastic Leukemia most commonly?
-
B-ALL: About 85% of cases, most common in children under 15 years. Peak incidence is below 3 years of age.
-
T-ALL: Presents in adolescents, usually as a thymic mass.
-
Boys are more commonly affected than girls.
-
ALL is the most common cancer of childhood.
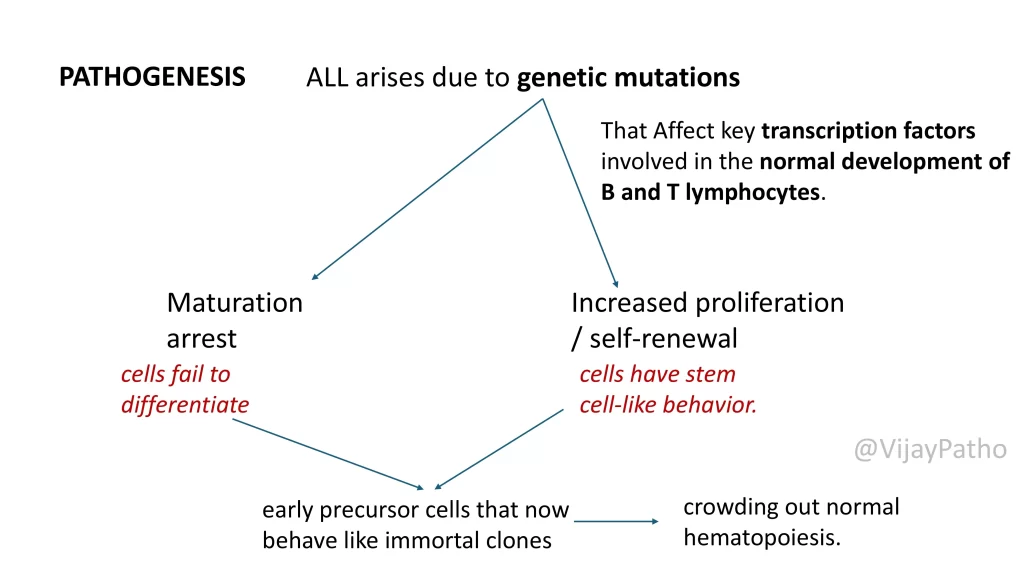
What causes Acute Lymphoblastic Leukemia ? (Pathogenesis)
The disease arises because of genetic mutations that affect transcription factors needed for normal B- and T-cell development.
Two key mechanisms:
-
Maturation arrest → cells cannot differentiate further.
-
Increased proliferation/self-renewal → cells behave like stem cells.
Together, these changes produce immortal lymphoblast clones that crowd the bone marrow and suppress normal blood cell formation.
Which genes are involved?
-
B-cell ALL: PAX5, TCF3, ETV6, RUNX1, BCR-ABL1 (Philadelphia chromosome), KMT2A, PBX1.
-
T-cell ALL: NOTCH1 gene mutation (critical for T-cell development).
Are single mutations enough to cause Acute Lymphoblastic Leukemia?
No. ALL is a multi-step process.
-
Initial mutations cause maturation arrest.
-
Additional driver mutations (like BCR-ABL1 fusion or RAS pathway mutations) push cells towards full leukemogenesis.
What about chromosomal abnormalities?
Seen in 90% of ALL cases.
-
Hyperdiploidy (>50 chromosomes): Good prognosis.
-
Hypodiploidy (<44 chromosomes): Poor prognosis.
-
Structural translocations:
-
ETV6-RUNX1 fusion
-
BCR-ABL1 fusion
-
TCF3-PBX1 fusion
-
These are important for diagnosis, risk stratification, and targeted therapy.
What happens in the bone marrow?
Accumulation of lymphoblasts → suppression of normal hematopoiesis →
-
Anemia (fatigue, pallor)
-
Neutropenia (fever, infections)
-
Thrombocytopenia (bleeding, petechiae)
Leukemic cells may also spread to blood, CNS, lymph nodes, liver, spleen, and testes.
What does Acute Lymphoblastic Leukemia look like morphologically?
-
Peripheral smear:
-
Anemia
-
Leukocytosis with blasts
-
Neutropenia
-
Thrombocytopenia
-
-
Bone marrow:
-
Hypercellular marrow packed with lymphoblasts
-
Lymphoblasts: large cells, scant basophilic cytoplasm, delicate nuclear chromatin, small nucleoli, sometimes convoluted nuclei
-
-
Histopathology:
-
Classic “starry sky” pattern due to macrophages eating apoptotic cells
-
How do you differentiate ALL from AML?
-
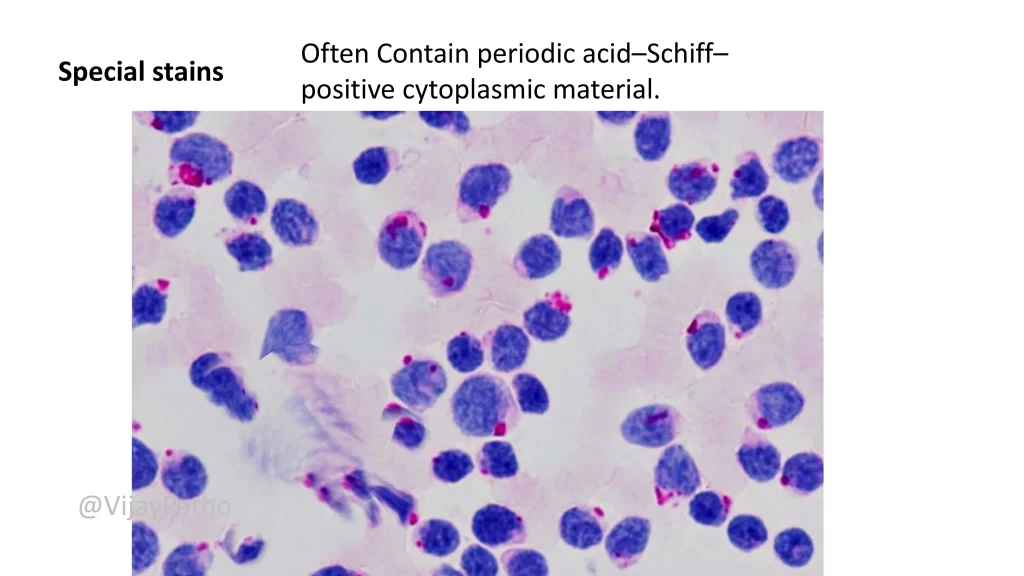
Morphology may be tricky.
-
Special stain: Periodic Acid–Schiff (PAS) shows block positivity in lymphoblasts.
-
Definitive diagnosis: Flow cytometry
-
B-ALL: CD19, CD22, CD10, TdT positive
-
T-ALL: CD1, CD2, CD5, CD7 positive
-
Click to read more about Cytochemical stains in Hematolymphoid neoplasms
What are the clinical features of Acute Lymphoblastic Leukemia ?
-
Abrupt onset with stormy course
-
Marrow failure signs:
-
Fatigue (anemia)
-
Fever (neutropenia)
-
Bleeding (thrombocytopenia)
-
-
Bone pain (marrow expansion, periosteal infiltration)
-
Lymphadenopathy, hepatosplenomegaly, testicular enlargement
-
CNS involvement: headache, vomiting, nerve palsies (more common in ALL than AML)
How is Acute Lymphoblastic Leukemia treated?
-
Aggressive chemotherapy
-
95% of children achieve remission
-
75–85% are cured
-
-
This is why it is very important to distinguish ALL from AML, as AML is usually not curable with chemotherapy alone.
What are the prognostic factors in Acute Lymphoblastic Leukemia ?
Favorable prognosis:
-
Age 2–10 years
-
Low WBC count
-
Hyperdiploidy (>50 chromosomes)
-
Extra copies of chromosomes 4 and 10
-
Translocation t(12;21) → ETV6-RUNX1 fusion
Poor prognosis:
-
Age <2 years or adolescence/adulthood
-
CNS involvement
-
Very high WBC count (>100,000/µL)
-
Hypodiploidy
Can ALL benefit from targeted therapy?
Yes.
-
Patients with tyrosine kinase mutations (e.g., Philadelphia chromosome with BCR-ABL1) can receive targeted therapy (TKIs) along with chemotherapy
Click below to watch the video on Acute Lymphoblastic Leukemia